
The major drawback of MS/MS, however, is that fragmentations within the thioether rings do not result in new fragment ions and hence cannot be used to accurately decipher ring patterns ( Lohans & Vederas, 2014). Tandem MS methodologies have also proven useful in the characterization of ring patterns of lanthipeptides. 
Therefore, mass spectrometric data should always be paired with chemical derivatization assays and/or NMR spectroscopy if sufficient amounts of materials can be obtained. This mass shift can also be indicative of the presence of Lan and MeLan rings in the mature lanthipeptide. For example, the most typical post-translational modification introduced into lanthipeptides involves a loss of water corresponding to a mass shift of − 18 Da per dehydrated Ser/Thr (Dha/Dhb). The difference in monoisotopic mass between the theoretical unmodified amino-acid sequence and the mature lanthipeptide provides valuable insight into the number and nature of post-translational modifications imparted to the core peptide. van der Donk, in Methods in Enzymology, 2021 6.6.1 OverviewĬomparison of the monoisotopic masses of a mature lanthipeptide and precursor peptide can be useful for structural elucidation. As the atomic number increases, the atomic mass increases as well.Richard S. The mass of an atom is equal to the sum of its protons and neutrons. As a result, the atomic number grows with each element. The atomic number forms the base for the periodic table. Is atomic mass always proportional to atomic number? The atomic number of an element is used to represent the number of protons in its nucleus.ĥ. The amount of neutrons and protons in an element's nucleus determines its atomic mass. How do atomic number and atomic mass number relate to one other? 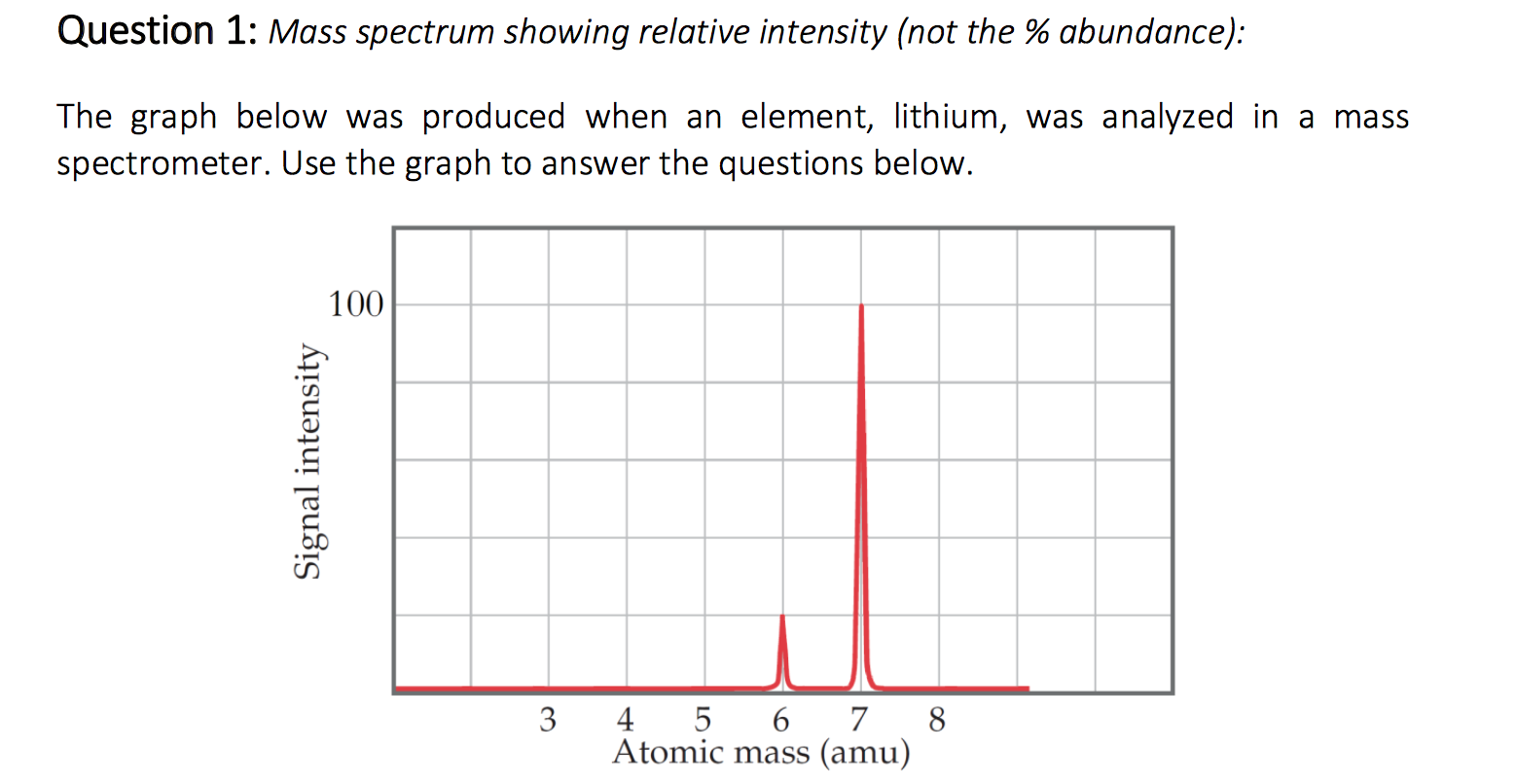
To find out how many atoms are in a sample, divide its weight in grammes by the AMU atomic mass from the periodic table, then multiply by Avogadro's number: 6.02 x 10^23Ĥ. How do you calculate the mass of an atom? Protons, neutrons, and electrons are the particles that makeup atoms and are responsible for their mass and charge.ģ. An element's average atomic mass is computed by adding the masses of its isotopes, each multiplied by its natural abundance on Earth. Isotopes are variants of an element with various neutrons that have different masses. To determine the atomic mass, what average do you need?
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
